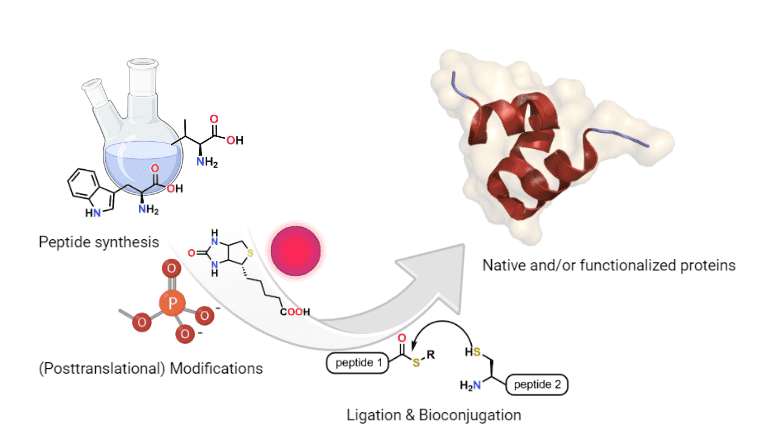
In organic chemistry, peptide synthesis refers to the chemical production of peptides through the formation of amide (peptide) bonds between amino acids.
This process involves a condensation reaction between the carboxyl group of one amino acid and the amino group of another, typically requiring protecting group strategies to prevent side reactions involving reactive side chains. Chemical synthesis generally proceeds from the C-terminus to the N-terminus, which is opposite to the direction of ribosomal protein biosynthesis in living cells.
Although traditional solution-phase synthesis was initially used, it has largely been replaced in research and development by solid-phase peptide synthesis (SPPS) due to improved efficiency and automation. However, solution-phase methods remain valuable for industrial-scale production of small peptides. Chemical synthesis enables the generation of sequences that are difficult to express biologically, as well as the incorporation of non-natural amino acids, backbone modifications, and D-amino acids for enhanced stability and functionality.


So What Are Peptide Bonds?
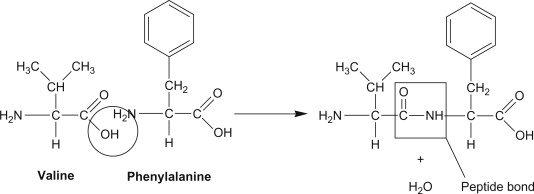
A peptide bond is a covalent amide linkage formed between the carboxyl group of one amino acid and the amino group of another. This condensation reaction releases a molecule of water and generates a –CO–NH– bond. Peptide bonds are the fundamental chemical connections that assemble amino acids into peptides and proteins.
Two common examples of dipeptides formed through peptide bonds are L-phenylalanyl-L-valine and L-leucyl-L-alanine, where the specific sequence of amino acids determines the structure and biological function of the resulting molecule.
Development of Solid-Phase Peptide Synthesis (SPPS)
Emil Fischer first introduced the concept of peptides in 1901, laying the foundation for stepwise peptide assembly. Early solution-phase synthesis advanced with the development of reversible protecting groups such as the Cbz group by Max Bergmann and Leonidas Zervas. While solution methods remain valuable for large-scale production, peptide synthesis became significantly more practical with the introduction of solid-phase peptide synthesis (SPPS).
Robert Bruce Merrifield revolutionized the field in the 1960s by attaching the growing peptide chain to a polystyrene resin, allowing excess reagents to be washed away easily. His original strategy used Boc chemistry and acid-mediated cleavage, enabling automated peptide synthesis and even total synthesis of proteins such as Ribonuclease A and enzymes related to HIV-1.
The later introduction of Fmoc chemistry by Louis A. Carpino provided a milder, base-labile alternative to Boc protection. Fmoc-based SPPS reduced acid-induced side reactions and significantly improved yields for sensitive, tryptophan-rich peptides such as Gramicidin A. Over time, methodological refinements allowed efficient synthesis of complex proteins including ubiquitin and HIV-related proteins.
By the mid-1990s, Fmoc chemistry had largely replaced Boc methods in research laboratories due to improved peptide quality, milder cleavage conditions, and better side-chain protection strategies. Today, SPPS is highly automated and widely used. Ongoing developments focus on synthesizing peptides containing non-natural amino acids, post-translational modifications, and complex biomolecular architectures, supported by advances in solid supports, coupling reagents, and analytical techniques.
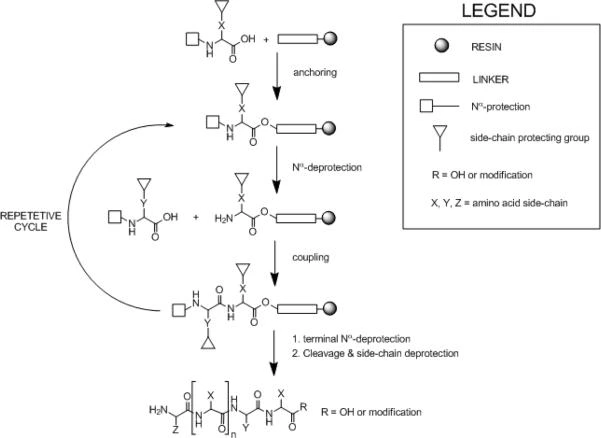
The Solid Support
Efficient solvation of the peptide resin complex is the most critical factor for successful chain elongation in solid-phase peptide synthesis. Swollen resin beads can be handled in batch mode with agitation and filtration, or arranged in columns for continuous-flow synthesis. Under optimal solvation, the polymer matrix becomes highly accessible to reagents, promoting efficient coupling reactions.
NMR studies (¹H, ²H, ¹³C, ¹⁹F) have shown that solvent-swollen polystyrene (PS) resins behave almost like polymers in solution, allowing reagents to access reactive sites effectively. Similar mobility has been observed in Pepsyn resins. To further enhance solvation, modified supports such as PEG-grafted polystyrene (PEG-PS) and polyoxyethylene–polystyrene (POE-PS) were developed, combining the homogeneous reaction environment of liquid phase chemistry with the practical advantages of an insoluble solid support. Other advanced supports including PEGA (polyethylene glycol polyacrylamide), CLEAR (cross-linked acrylate ethoxylate resin), and ASPECT (augmented surface polyethylene) offer improved swelling properties and broader applicability, particularly in organic and combinatorial synthesis.
Difficult peptide sequences can often be synthesized successfully by adjusting the solid support conditions. As peptide chains grow longer, the peptide resin complex becomes increasingly polar, potentially reducing coupling efficiency. This issue can be mitigated by modifying solvent systems, incorporating polar or chaotropic additives, or using resins with lower substitution levels to minimize steric crowding. Additionally, in Fmoc based synthesis, slow deprotection steps can be monitored spectrophotometrically, allowing optimization of base treatment times and solvation conditions to ensure complete reactions.

Coupling Reagents in Solid-Phase Peptide Synthesis (SPPS)
Coupling reagents are essential for activating the carboxyl group of an amino acid to enable peptide bond formation. The classical in situ reagents include N,N′-dicyclohexylcarbodiimide (DCC) and N,N′-diisopropylcarbodiimide (DIC), which promote amide bond formation between protected amino acids. However, carbodiimide mediated reactions are significantly improved by additives such as 1-hydroxybenzotriazole (HOBt) and 1-hydroxy-7-azabenzotriazole (HOAt). These additives accelerate coupling, suppress racemization, and prevent dehydration side reactions of asparagine (Asn) and glutamine (Gln) side chains.More reactive phosphonium and uronium based reagents were later developed, including BOP, PyBOP, PyAOP, HBTU, HATU, and TBTU. These reagents provide faster coupling kinetics than carbodiimides and are widely used in modern SPPS protocols due to their efficiency and reduced side reactions.
Amino acid halides have also proven valuable. Fmoc–amino acid chlorides and fluorides react rapidly under SPPS conditions, especially in the presence of HOBt and bases such as DIEA (N,N-diisopropylethylamine), with minimal racemization. The reagent tetramethylfluoroformamidinium hexafluorophosphate (TFFH) enables convenient preparation of Fmoc amino acid fluorides for automated synthesis. Amino acid fluorides are particularly advantageous for incorporating sterically hindered residues, such as those found in peptaibols, improving overall coupling efficiency.
Today, these coupling reagents and additives are commercially available and form the backbone of reliable, high-efficiency peptide synthesis methodologies.
Protein Synthesis
Chemical protein synthesis relies on three main strategies: stepwise synthesis, where the entire protein is assembled sequentially amino acid by amino acid; fragment assembly, in which shorter peptide segments are synthesized, purified, and then covalently linked; and directed assembly, where prepared peptide strands associate noncovalently into protein-like structures. Fragment assembly can involve either convergent coupling of protected fragments or chemoselective ligation of unprotected peptides, most notably through native chemical ligation, developed by Stephen B. H. Kent and Philip E. Dawson, which joins a peptide thioester to an N-terminal cysteine to form a native amide bond under mild conditions. Together, these complementary approaches enable the efficient construction of complex and modified proteins beyond the limits of traditional stepwise synthesis alone.