Antimicrobial Peptides (AMPs)
Evolution-Driven Applications in Modern Antimicrobial Strategies
Introduction to Antimicrobial Peptides
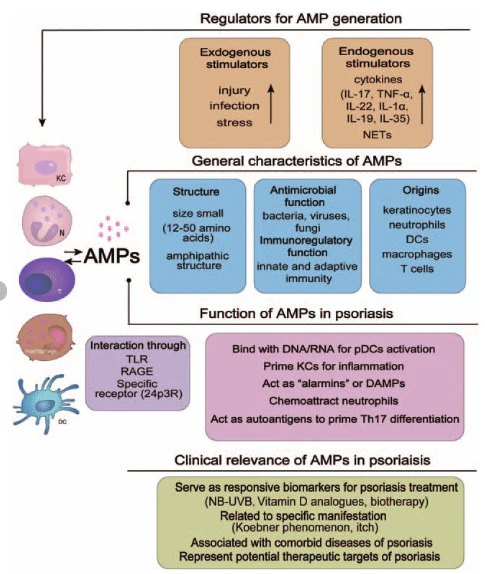
Antimicrobial peptides (AMPs) are short, biologically active proteins that play a critical role in innate immunity. They exhibit broad-spectrum antimicrobial activity against bacteria, viruses, and fungi. Often referred to as host-defense peptides, AMPs are naturally present in epithelial tissues and are also produced systemically in response to infection in multicellular eukaryotes.
AMPs are widely distributed across plant and animal species, reflecting their fundamental role in defense mechanisms. Most genomes encode multiple AMP gene families, typically ranging from 5 to 10 families, each containing several paralogous genes. This genetic diversity highlights their evolutionary importance and functional adaptability.
Currently, more than 3,000 antimicrobial peptides have been identified from various biological sources. Their strong antimicrobial potential, combined with increasing global concerns about antibiotic resistance, makes AMPs promising candidates for the development of next-generation antimicrobial drugs, surface disinfectants, and therapeutic agents. Many AMP-based compounds are already undergoing clinical evaluation.

Structural and Functional Characteristics of AMPs
AMPs are characterized by their relatively small size, typically ranging from 15 to 150 amino acids. A defining feature is their net positive charge, which enables them to interact with negatively charged microbial membranes. Structurally, AMPs contain both hydrophilic and hydrophobic regions, allowing them to function effectively in aqueous and lipid environments.
This amphipathic nature is essential for their antimicrobial activity. It facilitates membrane binding, insertion, and disruption. Due to their simple biochemical structure, AMPs can evolve rapidly and independently across different species. For example, defensins—a well-known class of AMPs—have evolved multiple times in plants, insects, and vertebrates, demonstrating convergent evolution.
Classical Mechanisms of Antimicrobial Action
Traditionally, AMPs were thought to act primarily by disrupting microbial cell membranes. After binding to the membrane, they insert into the lipid bilayer, forming pores that compromise membrane integrity. This leads to leakage of cellular contents and ultimately cell death.
However, research has revealed that AMP activity is more complex. In addition to membrane disruption, AMPs can:
- Inhibit intracellular processes such as protein synthesis and metabolism
- Interfere with nucleic acid functions
- Form extracellular nanonets that trap microbes
- Target specific intracellular proteins after entering the cell
These multiple mechanisms enhance their antimicrobial effectiveness and reduce reliance on a single mode of action.
Synergistic Interactions Among AMPs
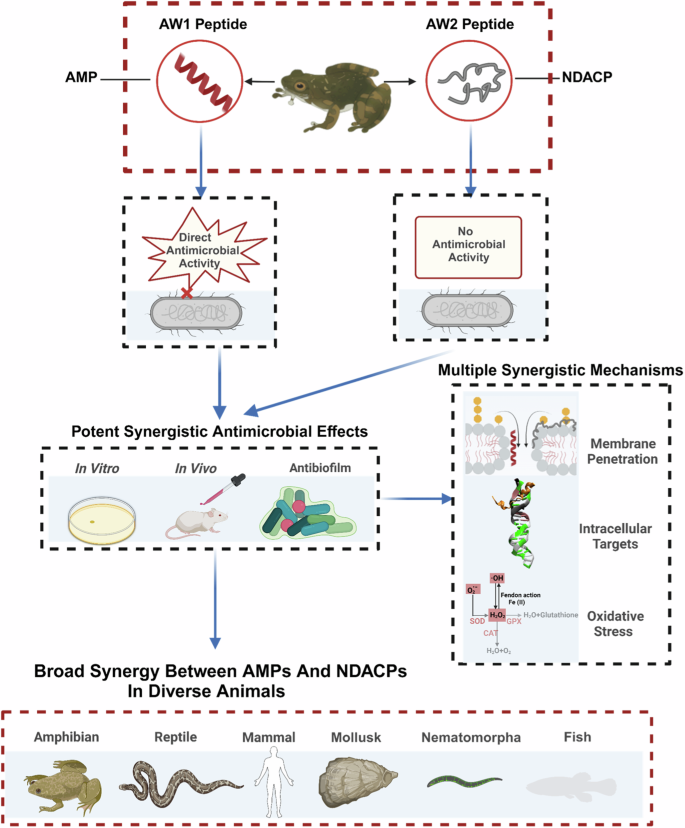
One of the most important properties of antimicrobial peptides is their ability to act synergistically. Different AMPs can work together to enhance antimicrobial efficacy. For example, one peptide may disrupt the membrane, allowing another peptide to enter the cell and target intracellular components.
This synergistic behavior significantly increases antimicrobial efficiency and reduces the required concentration of individual peptides. It also plays a key role in limiting the development of microbial resistance.
In natural biological systems, organisms often produce multiple AMPs simultaneously in response to infection. This coordinated response ensures broad and effective protection against diverse pathogens.
Revisiting the Classical Model of AMP Function
Earlier models suggested that AMP activity was largely nonspecific and dependent on reaching a threshold concentration. According to this view:
- All AMPs were functionally interchangeable
- Higher concentrations ensured effective microbial killing
- Resistance evolution was unlikely due to the simplicity of action
However, recent research challenges this perspective. Evidence now shows that AMPs can exhibit high specificity toward particular pathogens. Individual peptides may be critical for defense against specific microorganisms, rather than acting as part of a redundant system.
This shift in understanding highlights the importance of peptide identity, not just concentration, in determining antimicrobial effectiveness.
Evolutionary Diversification and Specificity
AMP genes evolve rapidly through processes such as gene duplication, mutation, and deletion. This dynamic evolution allows organisms to adapt to changing microbial environments.
New findings indicate that:
- Specific AMPs target specific pathogens
- Small genetic variations can significantly alter antimicrobial activity
- Natural selection drives diversification based on ecological pressures
In some cases, identical or similar AMP variants have evolved independently in different species, demonstrating convergent evolution. Additionally, polymorphisms within species suggest that multiple peptide variants may be maintained to combat diverse microbial threats.
Role of AMPs in Host–Microbe Interactions
Beyond pathogen defense, AMPs play a crucial role in regulating beneficial microbial communities. In the gut and other tissues, they help maintain a balanced microbiota, which is essential for overall health.
Functions include:
- Controlling microbial population density
- Preventing overgrowth of harmful bacteria
- Supporting symbiotic relationships
AMPs are also involved in highly specialized symbiotic systems. For example, certain insects and marine organisms use AMPs to regulate beneficial bacteria that provide essential metabolic functions.
Interestingly, in some cases, AMPs are produced at sublethal concentrations. Instead of killing microbes, they modify bacterial physiology, enabling controlled interactions between host and symbionts.
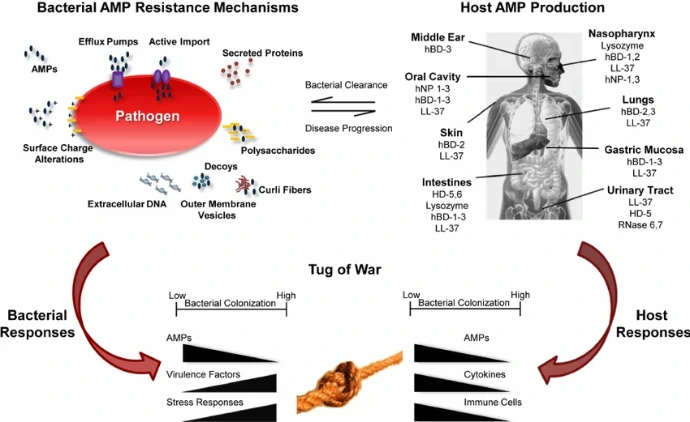
Resistance to Antimicrobial Peptides
Although AMPs were initially believed to be resistant-proof, it is now clear that microorganisms can develop resistance. However, resistance mechanisms are generally less specific and include:
- Modification of cell membrane charge
- Production of protective biofilms
- Activation of efflux pumps
- Secretion of degrading enzymes
Despite this, resistance to AMPs tends to evolve more slowly compared to conventional antibiotics. Several factors contribute to this:
- Rapid killing action reduces mutation opportunities
- Multiple targets limit adaptation pathways
- Synergistic interactions increase effectiveness
The pharmacodynamic properties of AMPs also play a role. They typically exhibit steep dose-response curves, meaning there is a narrow concentration window where resistance can develop.
Clinical and Therapeutic Applications
AMPs are increasingly being explored for medical and industrial applications. Their potential uses include:
- Treatment of multidrug-resistant infections
- Development of topical antiseptics
- Co-therapy with existing antibiotics
- Biofilm disruption in chronic infections
One promising approach is combining AMPs with conventional antibiotics. This strategy can restore the effectiveness of drugs that have lost activity due to resistance.
Additionally, host-directed therapies aim to stimulate the body’s natural AMP production. This approach enhances immune response without directly targeting pathogens, reducing the risk of resistance development.
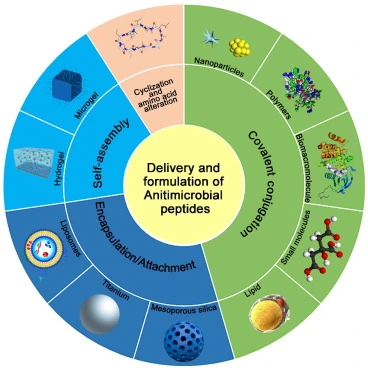
Synthetic biology also enables the design of engineered peptides with improved stability, specificity, and activity. Hybrid and chimeric peptides are being developed to target high-risk pathogens.
Challenges in AMP Development
Despite their potential, several challenges must be addressed:
- Differences between in vitro and in vivo activity
- Stability and degradation in biological environments
- Potential toxicity at high concentrations
- Risk of cross-resistance with natural host peptides
Standard laboratory tests, such as minimum inhibitory concentration (MIC), may not accurately reflect real biological conditions. Therefore, more advanced models are needed to evaluate AMP performance in complex environments.
Conclusion
Antimicrobial peptides represent a powerful and versatile class of natural defense molecules with significant potential in modern medicine. Their diversity, rapid evolution, and ability to function in synergistic networks make them highly effective against a wide range of pathogens.
Recent advances have shifted the understanding of AMP function from nonspecific, redundant agents to highly specialized and adaptive molecules. This new perspective emphasizes the importance of evolutionary context in designing AMP-based therapies.
By leveraging their natural properties and avoiding past mistakes associated with antibiotic misuse, AMPs can play a key role in addressing the global challenge of antimicrobial resistance.